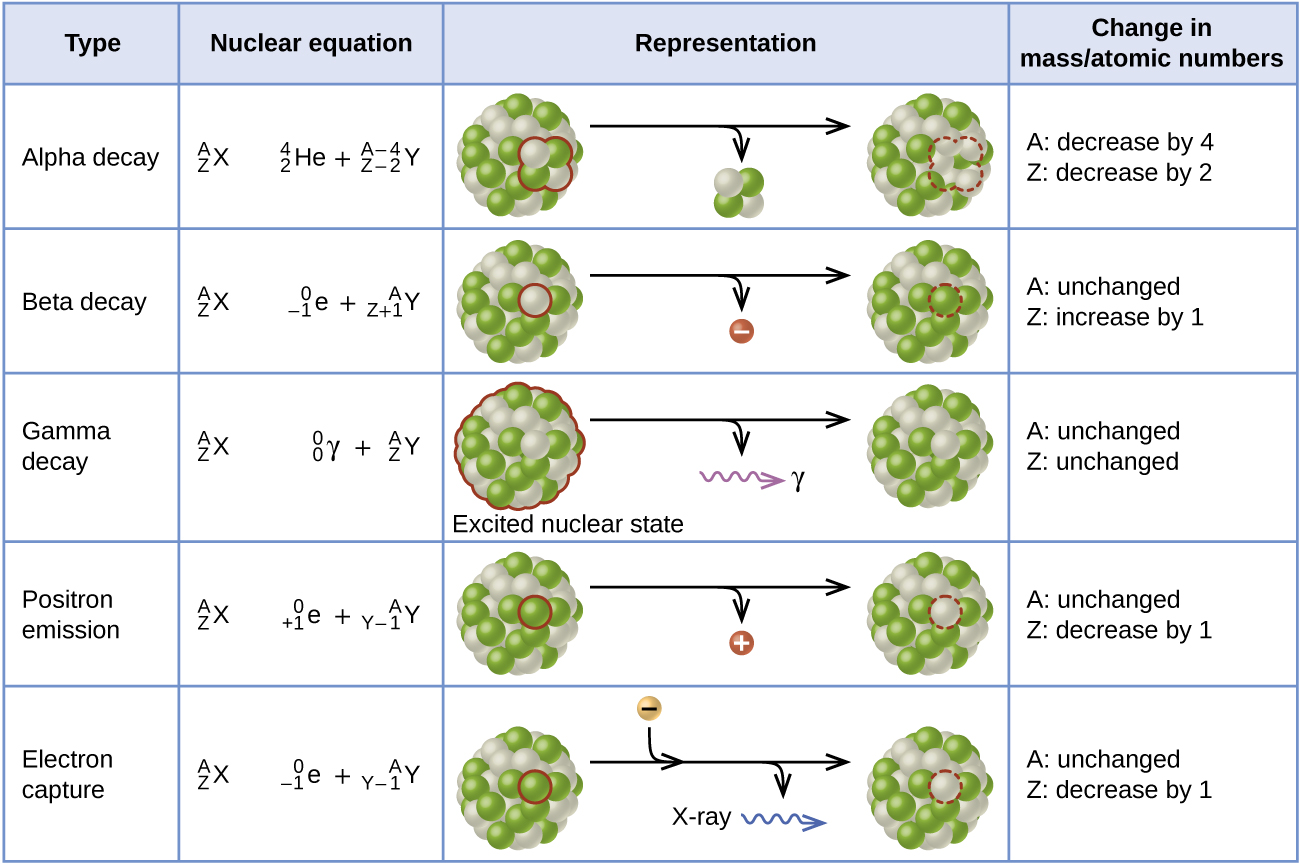
Since the gamma rays are in substance only a very high-energy photons, they are very penetrating matter and are thus biologically hazardous. As was written, they are produced by the decay of nuclei as they transition from a high energy state to a lower state. Gamma rays from radioactive decay are in the energy range from a few keV to ~8 MeV, corresponding to the typical energy levels in nuclei with reasonably long lifetimes. Gamma rays are high-energy photons with very short wavelengths and thus very high frequency. 
Typically after a beta decay (isobaric transition), nuclei usually contain too much energy to be in its final stable or daughter state. In most practical laboratory sources, the excited nuclear states are created in the decay of a parent radionuclide, therefore a gamma decay typically accompanies other forms of decay, such as alpha or beta decay. We usually indicate isomers with a superscript m, thus: 241mAm or 110mAg. Note that, nuclides with equal proton number and equal mass number (thus making them by definition the same isotope), but in a different energy state are known as nuclear isomers. This transition ( γ decay) can be characterized as:Īs can be seen, if a nucleus emits a gamma ray, atomic and mass numbers of daughter nucleus remain the same, but daughter nucleus will form different energy state of the same element. Gamma decay or γ decay represents the disintegration of a parent nucleus to a daughter through the emission of gamma rays (high energy photons).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
